
NEWS
新闻中心
SERVERS
服务项目
发布时间:2023-01-31
原文献:Ali, Z., Vildevall, M., Rodriguez, G.V. et al. Zebrafish patient-derived xenograft models predict lymph node involvement and treatment outcome in non-small cell lung cancer. J Exp Clin Cancer Res 41, 58 (2022).

癌症患者在治疗效果和肿瘤侵袭性方面表现出高度的个体差异,而这两方面恰恰是影响肿瘤患者生存的关键所在,因此,准确预测肿瘤转移风险和治疗结果对个体化治疗至关重要[1]。小鼠患者源性异种移植(PDX)模型在预测治疗结果方面表现出很高的准确性,但预测肿瘤侵袭性的方法仍然缺乏。另外,小鼠PDX实验周期较长,难以满足临床个体化治疗的需求[2-4]。
近年来,斑马鱼肿瘤异种移植(zPDX)系统已成为肿瘤学和肿瘤生物学研究中一种强有力的体内补充系统[5,6]。通过移植肿瘤细胞系来生成zCDX模型,已经使我们对驱动早期肿瘤扩散的分子和病理生理事件[7,8]、肿瘤微环境内的相互作用[9,10]以及对药物或治疗的反应[11-13]有了更深入的理解。由于已建立的细胞系可能随着时间的推移而发生表型变化,这些模型主要用于基础研究或早期临床前药物开发。为了更好地保持患者肿瘤的特征,研究者成功构建了直接移植患者来源的肿瘤样本的zPDX模型,并证明该模型能够准确地反应相应患者的肿瘤特性[14-19]。
本研究开发了一种新的、可靠的方法,成功将非小细胞肺癌(NSCLC)组织植入到斑马鱼幼鱼中建立zPDX模型。该zPDX模型准确地再现了相应小鼠PDX模型和患者自身对紫杉醇和厄洛替尼的反应。重要的是,该zPDX平台预测肿瘤向患者淋巴结扩散的敏感性为91%,而预测淋巴结无侵犯的特异性为62%。
● 主要研究结果
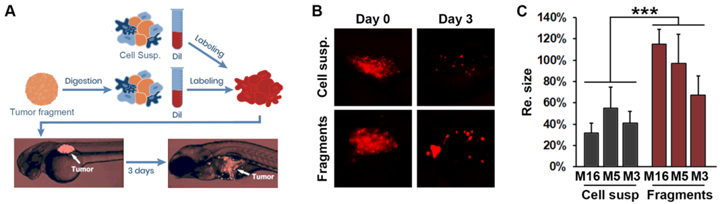
1. 对比冻存的组织与单细胞悬液,研究结果显示冻存的患者组织移植成功率优于冻存的患者单细胞悬液,组织移植成功率高于80%(图1)。
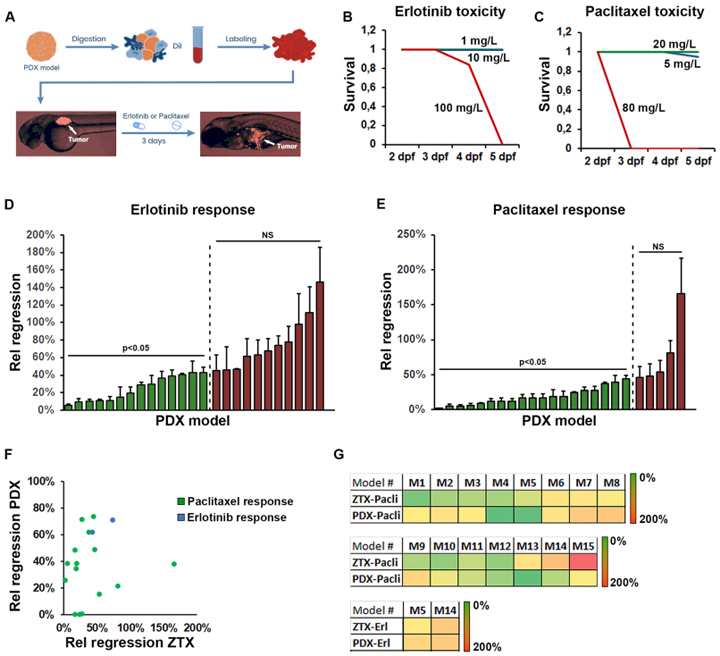
2、用25例zPDX模型评价厄洛替尼和紫杉醇的肿瘤治疗效果,结果显示厄洛替尼和紫杉醇在zPDX上对NSCLC肿瘤的治疗有效率与真实世界中患者的治疗有效率相似(图2D、E)。与药物评价的金标准小鼠PDX模型对比,zPDX对厄洛替尼和紫杉醇的应答与小鼠PDX高度一致(图2F、G)。
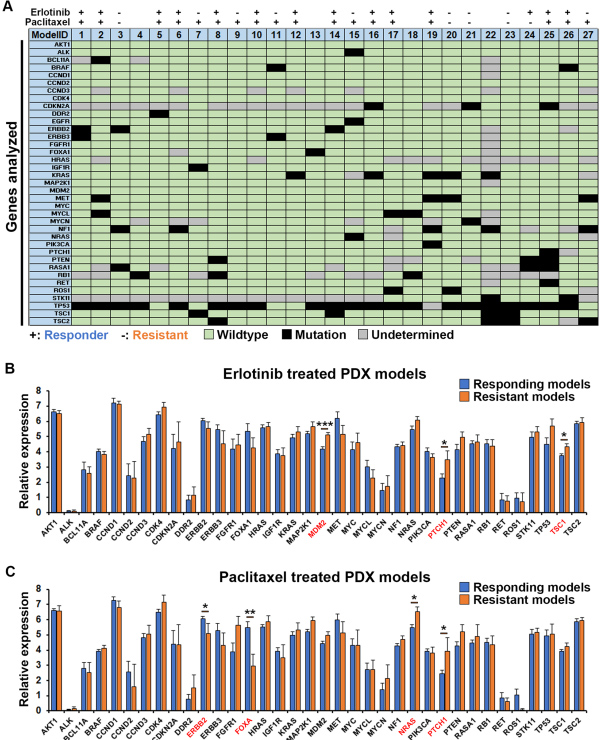
3. 将肿瘤组织进行测序分析,对比与药物治疗相关的靶点后发现,厄洛替尼和紫杉醇在zPDX上的治疗效果与基因检测结果相似(图3)。
4、将发生转移和未发生转移患者的肿瘤组织移植至斑马鱼,实验结果显示zPDX模型预测肿瘤向患者淋巴结扩散的敏感性为91%,而预测淋巴结无侵犯的特异性为62%(图4),提示zPDX模型可准确预测患者体内肿瘤转移的发生。

环特生物作为斑马鱼技术应用领导者,深度挖掘、创新斑马鱼技术的应用场景,在原有斑马鱼CDX模型用于抗肿瘤药物药效评价的基础上,成功建立多种实体瘤斑马鱼PDX药敏评价体系,实现临床应用,为患者提供精准的用药指导。同时,我们积极进行基于真实世界肿瘤患者的临床研究,联合浙江大学附属第二医院开展全世界首个胃癌斑马鱼PDX与患者治疗效果的一致性评价项目(Clinical Trials注册号:NCT05616533,详情点击:喜报丨浙大二院与环特生物合作的斑马鱼PDX临床项目通过美国临床研究注册(Clinical Trial)!),为斑马鱼PDX的临床应用提供更加充分的科学依据。
原文链接:https://doi.org/10.1186/s13046-022-02280-x
参考资料:
1. Eberhardt WE, et al. 2nd ESMO Consensus Conference in Lung Cancer: locally advanced stage III non-small-cell lung cancer. Ann Oncol. 2015;26:1573–88.
2. Hidalgo M, et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov. 2014;4:998–1013.
3. Schueler J, et al. Patient derived renal cell carcinoma xenografts exhibit distinct sensitivity patterns in response to antiangiogenic therapy and constitute a suitable tool for biomarker development. Oncotarget. 2018;9:30946–61.
4. Schueler J, et al. Induction of Acquired Resistance towards EGFR Inhibitor Gefitinib in a Patient-Derived Xenograft Model of Non-Small Cell Lung Cancer and Subsequent Molecular Characterization. Cells. 2019;8(7):740.
5. Rouhi P, et al. Hypoxia-induced metastasis model in embryonic zebrafish. Nat Protoc. 2010;5:1911–8.
6. Xiao J, Glasgow E, Agarwal S. Zebrafish Xenografts for Drug Discovery and Personalized Medicine. Trends Cancer. 2020;6:569–79.
7. Liu C, et al. A Zebrafish Model Discovers a Novel Mechanism of Stromal Fibroblast-Mediated Cancer Metastasis. Clin Cancer Res. 2017;23:4769–79.
8. Svensson S, et al. CCL2 and CCL5 Are Novel Therapeutic Targets for Estrogen-Dependent Breast Cancer. Clin Cancer Res. 2015;21:3794–805.
9. Vazquez Rodriguez G, Abrahamsson A, Jensen LD, Dabrosin C. Estradiol Promotes Breast Cancer Cell Migration via Recruitment and Activation of Neutrophils. Cancer Immunol Res. 2017;5:234–47.
10. Vazquez Rodriguez G, Abrahamsson A, Jensen LDE, Dabrosin C. Adipocytes Promote Early Steps of Breast Cancer Cell Dissemination via Interleukin-8. Front Immunol. 2018;9:1767.
11. He X, et al. Visualization of human T lymphocyte-mediated eradication of cancer cells in vivo. Proc Natl Acad Sci U S A. 2020;117:22910–9.
12. Kabakci Z, et al. Pharmacophore-guided discovery of CDC25 inhibitors causing cell cycle arrest and tumor regression. Sci Rep. 2019;9:1335.
13. Selvaraju K, et al. Cytotoxic unsaturated electrophilic compounds commonly target the ubiquitin proteasome system. Sci Rep. 2019;9:9841.
14. Fior R, et al. Single-cell functional and chemosensitive profiling of combinatorial colorectal therapy in zebrafish xenografts. Proc Natl Acad Sci U S A. 2017;114:E8234–43.
15. Gaudenzi G, et al. Patient-derived xenograft in zebrafish embryos: a new platform for translational research in neuroendocrine tumors. Endocrine. 2017;57:214–9.
16. Gaudenzi G, et al. Patient-derived xenograft in zebrafish embryos: a new platform for translational research in neuroendocrine tumors. Endocrine. 2017;57:214–9.
17. Wu JQ, et al. Patient-derived xenograft in zebrafish embryos: a new platform for translational research in gastric cancer. J Exp Clin Cancer Res. 2017;36:160.
18. Lin J, et al. A clinically relevant in vivo zebrafish model of human multiple myeloma to study preclinical therapeutic efficacy. Blood. 2016;128:249–52.
19. Costa B, et al. Developments in zebrafish avatars as radiotherapy sensitivity reporters - towards personalized medicine. EBioMedicine. 2020;51:102578.